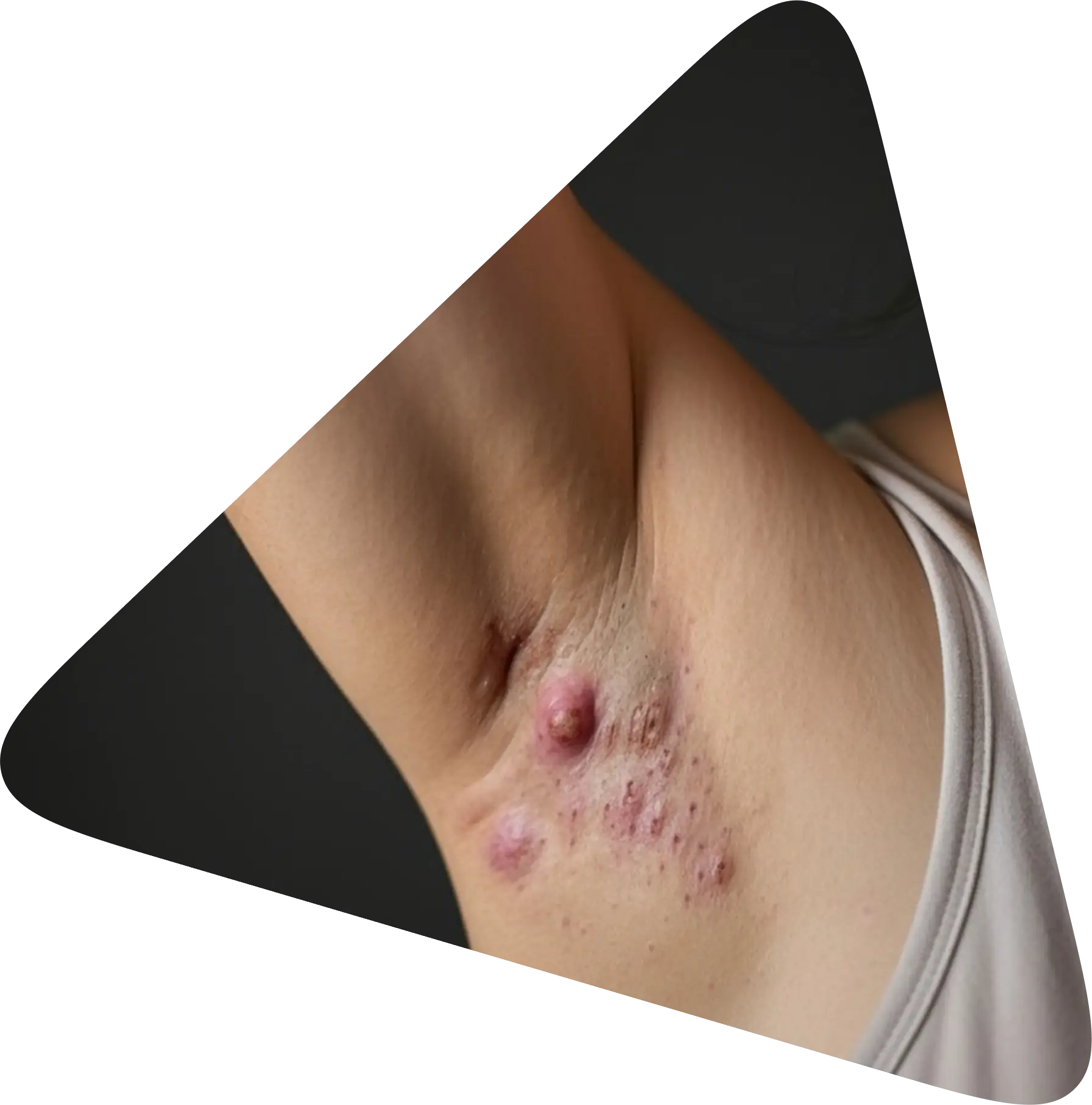
Clinical study on Hidradenitis Suppurativa
(Acne Inversa)
Do you suffer from recurring, painful inflammation in the armpits, groin, breasts or genital area? We are looking for patients to take part in a clinical study on chronic inflammatory and recurrent Hidradenitis Suppurativa, also known as Acne Inversa.
Potential participants must fulfil certain requirements in order to take part in the study.
Study information
Phase 2a
18+ years
Injection
Possible symptoms and consequences
of Hidradenitis Suppurativa
Symptoms
- Nodules and inflammation: painful, reddened skin nodules that often spread under the skin
- Abscesses: purulent inflammations that burst open and secrete foul-smelling secretions
- Fistula ducts: connecting ducts under the skin that link different centres of inflammation
- Itching, pain and a feeling of pressure
Consequences
- Chronic pain
- Restricted movement, especially in affected areas of the body near joints
- Scarring: repeated inflammation often results in hard, bulging scars
- Scar strands and skin distortions after years of progression
- Lymphatic drainage disorders when lymph nodes are affected
- Social isolation due to shame about the symptoms (e.g. odour, wounds)
- Depression and anxiety disorders
- Impairment of sexuality and partnership
- Incapacity for work in severe cases
Why the study is being conducted?
We are looking for participants for our study who suffer from the symptoms listed. The aim of the study is to evaluate the effect of the investigational drug to be tested on recurrent Hidradenitis Suppurativa and the change in quality of life for you as a patient. During the course of the study, you will receive comprehensive, no-cost and study-related treatment from our medical specialists.
Important information
Study treatment and time required
The patients are randomly divided into three groups. There are two different dosage forms. Group 1 receives dosage form 1, group 2 receives dosage form 2 and there is also a placebo group. The active substance is administered by injection under the skin. The study will be conducted in three phases:
- Preliminary examination: up to 6 weeks before the start of treatment (1 visit to the study site)
- Treatment phase: 12 weeks (9 visits, 6 injections)
- Follow-up care: 4 weeks (2 visits)
The total duration of the study is approximately 22 weeks and includes 12 visits to the study site. All participants are monitored by medical professionals throughout the course of the study and are examined at regular intervals.
What is Hidradenitis Suppurativa?
Hidradenitis Suppurativa (HS) is a chronic inflammatory skin disease that repeatedly causes painful inflammation in certain areas of the skin - especially where skin rubs against skin or where there are many sweat glands, e.g. in the armpits, groin area, under the breast or on the buttocks. The disease usually begins after puberty and progresses in episodes. Hidradenitis Suppurativa is not contagious.
Requirements for participation
You are suitable if you:
- are at least 18 years old
- have had moderate or severe Hidradenitis Suppurativa in at least two different parts of the body for at least 6 months
- have at least five abscesses or inflammatory nodules
- have had an inadequate response to oral antibiotics OR have developed a recurrence after stopping antibiotics OR have an intolerance OR have a contraindication to oral antibiotics for the treatment of your HS
Excluded from participation are persons who:
- have another skin disease in the treatment area
- are infected with hepatitis B, hepatitis C or the human immunodeficiency virus
- are currently pregnant, breastfeeding or planning to become pregnant in the near future
- have a history of severe allergies, non-allergic drug reactions or multiple drug allergies
- have recently participated in another clinical study
Participate in the study
FAQ on clinical studies –
Frequently asked questions
What are clinical studies for?

What are the advantages of a clinical study?

- Access to new therapies: Participants get early access to innovative treatments that are not yet widely available.
- Close monitoring: Participants receive intensive medical care during the study.
- Contribution to research: Participation helps to improve medical care for future patients.
- Possible alleviation of personal complaints: Depending on the study, the new therapy may also result in personal health benefits.
Can anyone take part in a study?

- Age, gender or health condition
- Existing medical conditions or medication
- Severity of the disease
How does a clinical study work?

- Preliminary examination: Medical examination to determine whether you are suitable for the study.
- Information session and consent: You will be informed in detail and give your written consent.
- Participation phase: Depending on the study, you will receive treatment, medication or an examination. Your state of health will be monitored regularly.
- Completion and follow-up: After the end of treatment, follow-up examinations may follow to observe long-term effects.
Is participation voluntary?

What benefit do I have?

- Personal benefit: Possible improvement in your state of health through new treatment approaches.
- Social benefit: You help to develop medicine for other patients.
- Medical care: Close medical supervision by the study team.